The Problem We Solve
The Problem We Solve
Clinical trial teams are under pressure to move faster but technology adoption often slows them down due to:
- Complex protocols translated poorly into digital systems
- eSource implementations delayed by limited internal expertise
- Site staff overwhelmed by admin, forms, and rework
- Data quality issues identified too late
The result: delayed startups, frustrated teams, and unnecessary operational risk.

Our Solution: Smarter Technology, Seamlessly Integrated

Nexa‑Trials removes friction from clinical trial setup and execution by embedding directly into your study operations.
We don’t replace your team. We extend it by providing the following services:
- Protocol‑driven eSource and eCRF design
- Advanced configuration and scripting
- Automated logic, validation, and data reuse
- Operational and back‑office support across the study lifecycle
Everything we build is designed to work the way your team already does. Just faster and smarter.

Our Services
End-to-End Clinical Trial Support Powered by Expertise

eSource Implementation
Nexa Trials delivers end-to-end implementation, configuration, training, and compliant integration for rapid and efficient trial adoption.
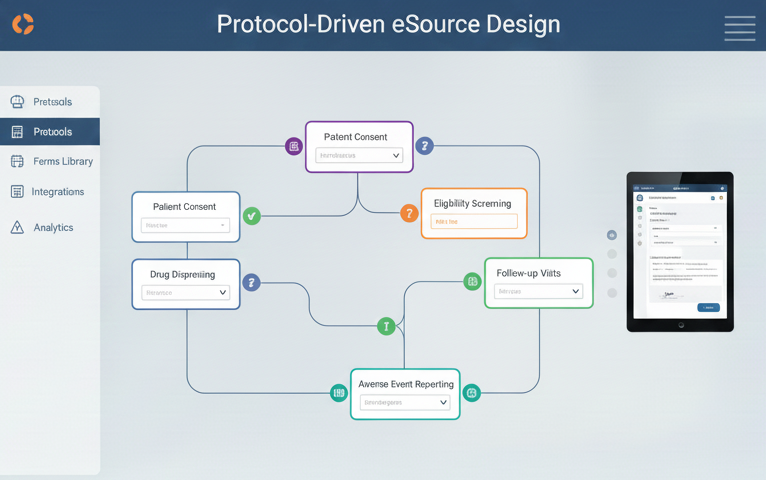
Protocol-Driven eSource Design
Protocol-Driven eSource Design tailored for accurate, compliant, and efficient digital data capture.
eSource Form Development
eSource forms optimized for accurate, compliant, and efficient digital data capture.

Customized JavaScripting
JavaScript methods to enable custom data validations, automated calculations (e.g. BMI, dosing, timepoints), and cross-visit coherence checks.

Data Entry, Cleansing & Migration
Data entry, cleansing, and migration services to transform legacy study data into clean, compliant, high-quality datasets ready for modern clinical systems.

Back-Office Operations Support
Back-office operations support, including study document generation, real-time reporting, visit tracking, issue resolution, and sponsor/site communication.

Who We Support

Clinical Research Sites
- Reduce administrative burden by up to 60%
- Improve staff adoption and satisfaction
- Spend more time on patient care, less on data entry

Sponsors & CROs
- Faster trial readiness and cleaner data
- Improved protocol adherence
- Reduced downstream queries and corrections
- Consistent data quality across studies
- Faster handovers and smoother inspections
Case Study

Digital Transformation at CenExel Anaheim

Over the last two years, Nexa Trials partnered with CenExel Anaheim to modernize their clinical research operations.
Key Outcomes:
- 80% of all trials now fully supported
- Faster and cleaner data collection
- Reduced operational burden
- Enhanced visibility for CRAs and sponsors
- Stronger compliance and audit readiness
Our collaboration helped CenExel Anaheim transition from manual workflows to a fully digital, real-time research environment.
Why Nexa Trials
Clinical‑First Approach: Built around real site operations
Deep Expertise: We handle complexity your team shouldn’t have to
Speed Without Compromise: Faster delivery without sacrificing compliance
Inspection‑Ready by Design: Built to withstand audits
How We Work
Assess: Review your protocol, timelines, and constraints
Design: Smart, compliant workflows tailored to your study
Build: Optimized configurations and automation
Support: Ongoing partnership throughout the trial lifecycle
Ready to Accelerate Your Next Trial?
Whether you’re planning a new study or struggling with an existing build, Nexa Trials helps you move faster with confidence.

