Clinical Trial Support Services
Nexa Trials partners with sponsors, CROs, and research sites to simplify complex clinical trial operations and accelerate study delivery. With deep expertise in protocol-driven eSource workflows, we help teams launch faster, capture cleaner data, and operate with confidence in regulated environments.

Implementation
Launch eSource quickly, correctly, and compliantly.
Implementing an advanced eSource platform requires more than technical setup—it demands clinical insight, operational understanding, and regulatory rigor. Nexa Trials ensures your eSource implementation delivers value from day one by:
- Configuring eSource to match your study protocol, workflows, and site realities
- Enabling rapid user adoption through structured training and hands-on support
- Integrating seamlessly with existing systems such as EHRs and laboratory platforms
- Ensuring full compliance with 21 CFR Part 11 and electronic record regulations
Our implementation expertise reduces startup risk, shortens timelines, and positions your study for long-term success.
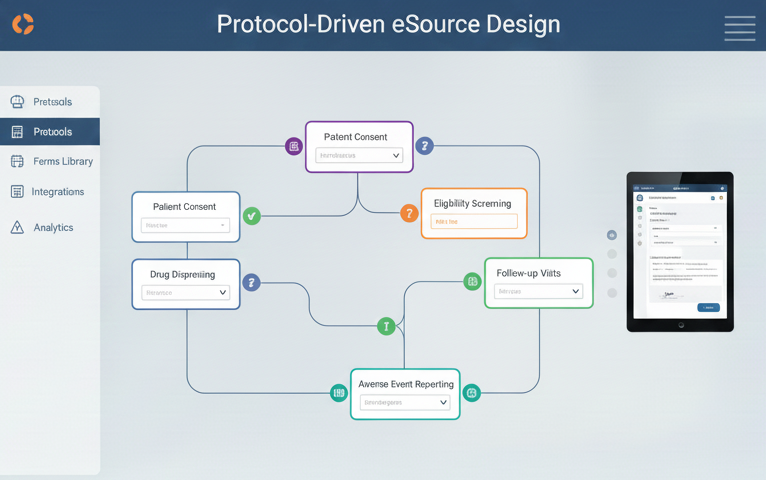
Protocol-Driven eSource Design
Turn complex protocols into site-ready digital workflows.
We transform detailed study protocols into intuitive, visit-aligned eSource designs that sites can execute efficiently and consistently.
- Clear conversion of protocol requirements into structured digital workflows
- Precise field mapping, visit schedules, and metadata configuration
- Advanced conditional logic to enforce protocol rules automatically
- Designs optimized for usability, efficiency, and high-quality data capture
The result: fewer ambiguities, fewer queries, and stronger protocol adherence across sites.
Custom Form Development
High-quality eCRFs built for performance, compliance, and usability.
Nexa Trials delivers end-to-end custom form development in eSource, tailored to your study’s unique requirements.
- Full lifecycle form development—from requirements and design to deployment
- Intelligent conditional logic, edit checks, and validations to prevent errors at entry
- Expert use of eSource built-in methods (e.g., findFormData, getItemDataContext) to enable dynamic behavior and data reuse
- Forms designed to align with CDISC/ODM standards, audit trails, and Part 11 compliance
- Mobile-friendly layouts and streamlined navigation to support busy site staff
We work closely with data managers, CRAs, and clinical teams to ensure every form supports both protocol intent and downstream analysis.
Ongoing support includes form updates, logic enhancements, metadata exports, and inspection-readiness support.

Advanced JavaScript Customization
Unlock the full power of eSource with tailored scripting.
Our JavaScript expertise enables sophisticated, study-specific behavior within eCRFs, improving accuracy and efficiency.
Capabilities include:
- Dynamic field behavior (show/hide, enable/disable based on subject data)
- Custom clinical validations (e.g., enforcing logical value relationships)
- Automatic calculations such as BMI, dose amounts, and time intervals
- Cross-form and cross-visit logic to maintain consistency across the study
These enhancements reduce manual review, improve data quality, and enhance the site experience.

Data Entry, Cleansing & Migration
Clean data you can trust—ready for analysis.
We support studies at every stage with reliable data services.
- Migration of legacy or external datasets into modern, compliant systems
- Data cleansing, reconciliation, and structural alignment
- Validation to ensure high-quality, analysis-ready datasets

Back-Office Operations Support
Operational support that keeps your study moving.
Nexa Trials acts as an extension of your study team, providing dependable operational support.
- Study document generation and maintenance
- Visit tracking, reconciliation, and operational oversight
- Real-time issue resolution and ongoing study support
- Sponsor and site communication assistance
